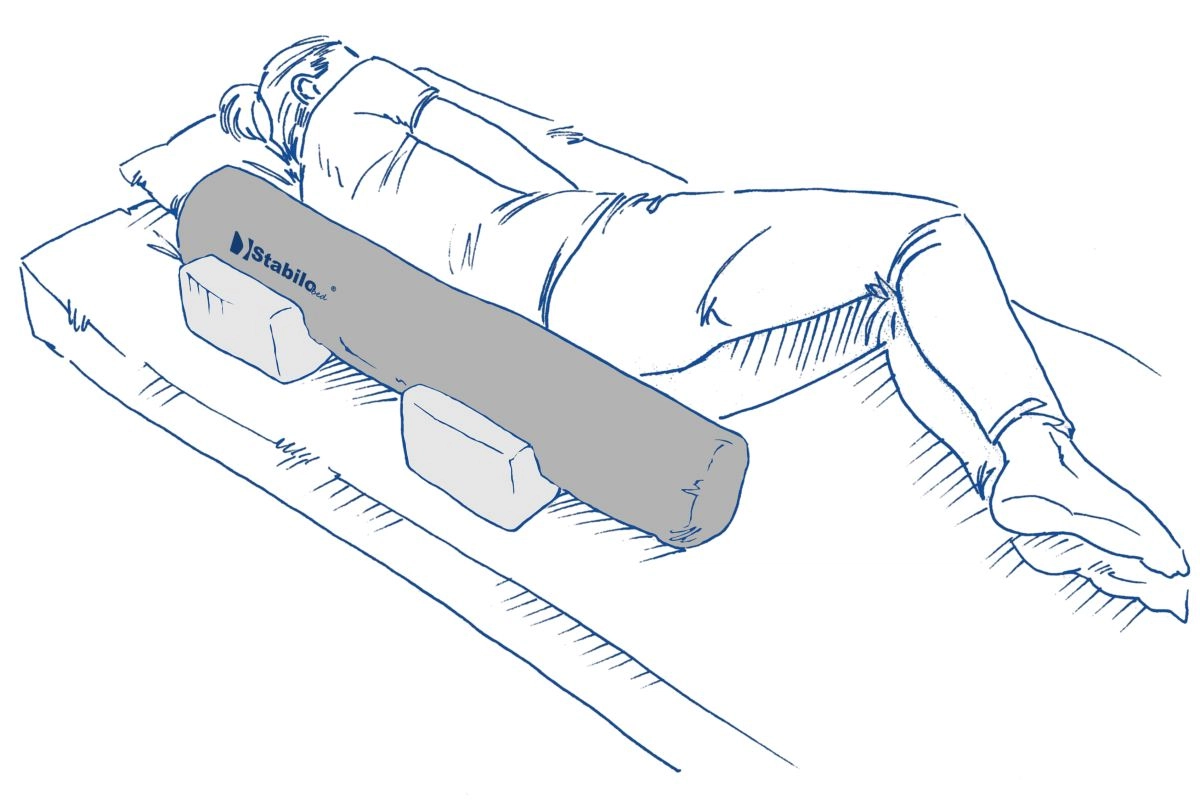
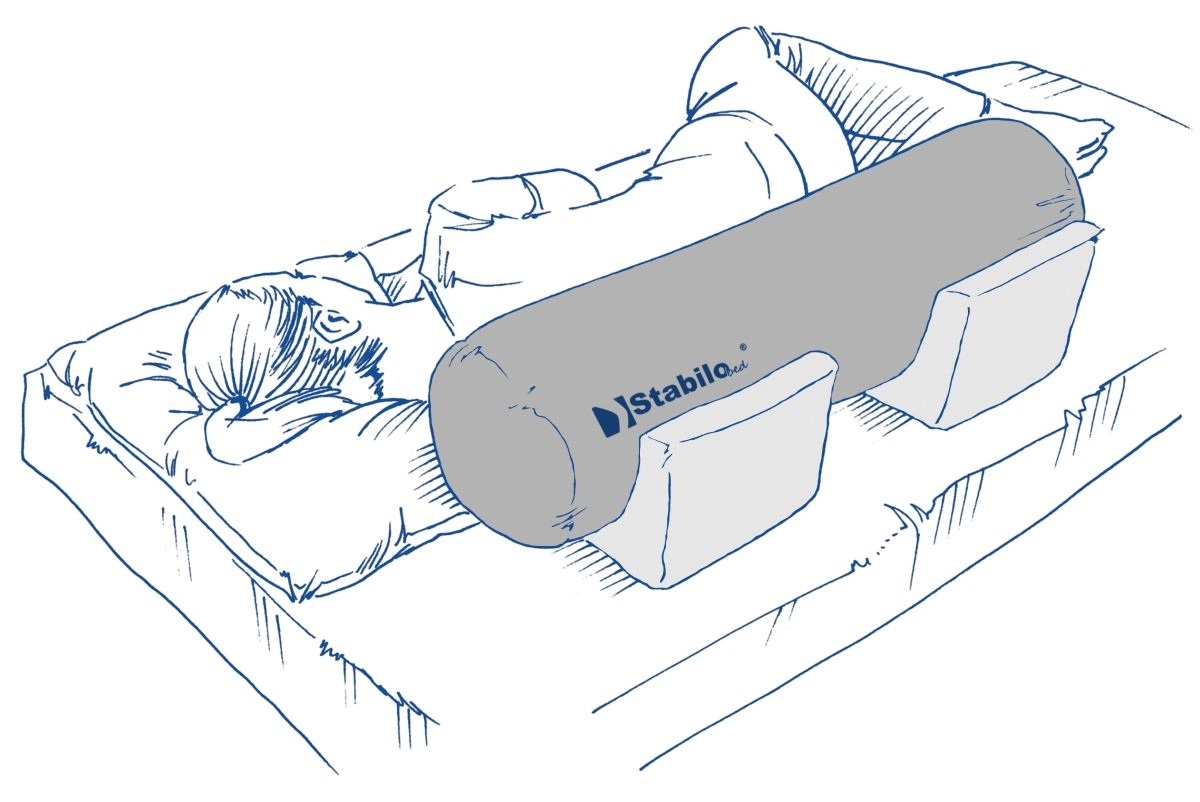
Description
Rehabilitation roller is made of rigid polyurethane foam. This special-shaped device is covered by durable, washable fabric with hygienic medical certificate. The rehabilitation rollers are mainly used in physiotherapy exercises. A wide range of available sizes allows to use them while massage therapy or user’s positioning therapy also. Our roller is 1st class medical device and it is easy to disinfect so it can be used by many users in health centres.
Structure and Maintenence
Structure

- PureMed™
- AirMesh™
- Comfort foam
- AntySlip™
Filling
PureMed™
Antibacterial fabric safe for people, easy to disinfect, and suitable for multiple users in hospitals or rehabilitation centers. Contains Silver Zeolite components and complies with REACH regulation. Light, soft and easy to shape.
AirMesh™
AirMesh is a special aerating-massaging fabric used in the places, which have a directis a special aerating-massaging fabric used in the places, which have a directcontact with the user’s body. As a result, the comfort of use has significantly increased, andat the same time the risk of bedsores has considerably decreased.
Comfort foam
Soft foam which has a lot of small, closed air bubbles. Because of that, it easily adjust to the user’s body shape.
AntySlip™
A special non-slip fabric which prevents the cushions and pillows from moving on the mattress.
Special features of PureMed™
- Compliant with the REACH regulation aimed at ensuring a high level of health and environmental protection.
- Passed the cigarette test and the matchstick test, proving it is a non-flammable fabric.
- Free of heavy metals, phthalates, allergic substances and carcinogens.
- Lightweight, soft, elastic and easy to shape.
- OEKO-TEX STANDARD 100® certified for skin-friendly parameters.
- Th fabric is Waterproof.
- Hygienic certificate issued by the National Institute of Public Health.
- The fabric is breathable.
- PureMed™ is vapour-permeable in one way only – the air from the inside may migrate freely outside.
- It is antibacterial and easy to disinfect.
Disinfection
Maintenance
PureMed™ meets all the essential requirements of the medical products of different kinds, according to the Regulation of the Ministers of Health on 3 November 2004 and the requirements contained in Annex I to Council Directive 93/42/EEC of 14 June 1993 concerning medical devices , deployed by the law of 30 April 2004 of Medical Devices (OJ L 93 of 2004., pos. 896). It is a flexible fabric which is safe for people. It is antibacterial, waterproof, vapour permeable and easy to disinfect.PureMed™ should be cleaned according to the laundry guide. Coated materials should be cleaned with the use of neutral pH soap solution (about 7) applied with a soft sponge or a damp cloth. Rub gently and wipe dry to remove the rest of the detergent. In order to keep the original colour and shine level, the procedure should be repeated every month.
Stains should come out if you use 25% solution of ethanol. Rub gently and wipe dry to remove the rest of the detergent.
Please, bear in mind that some stains might not be completely removed from the cleaned surface. It depends on several factors like active dyers permanently migrating to the material surface, the material composition and finish, or chemical composition of dirt as well as the time the soiling has remained on the material surface.
You should pay particular attention to the generally available domestic cleansers whose components might react with the soiling and cause an effect contrary to what was originally intended.
Remember that PureMed™ should never be cleaned with domestic cleansers and cleaning agents that are generally recommended for ‘eco-leather’.
PureMed™ Technical specification
| Surface weight |
200 ± 15 [g/m²] |
| Type of PU coating |
100 [%] |
| Type of PES carrier |
100 [%] |
| Strength breaking along |
≥20 [daN/5cm] |
| Strength breaking |
≥10 [daN/5cm] |
| Waterproof |
≥2000 [mm of H₂O] |
| Vapour permeability |
≥300 [g/m²/24h] |